Summary
Life sciences organizations face challenges when it comes to digital adoption, especially considering compliance, system complexity, and information silos. This is where digital transformation life sciences initiatives solve these challenges, helping to address these issues, with integration, governance, and analytics at the core.
What Are the Digital Adoption Challenges in Life Sciences – and How to Overcome Them?
Digital transformation in life sciences is challenging because every new system must fit into a tightly regulated ecosystem.
Companies working in pharmaceuticals, biotechnology, and medical research need to renew processes, make data more visible, and accelerate novel ideas. However, unlike other industries, the changes here should be cautious and gradual. Each system should be checked so that it is tracked and documented. And that is the reason why Digital transformation life sciences require a longer period for digital changes, as initially any careful approach is required.
In this blog, we examine the principal challenges life sciences organizations face in their adoption of digital tools and some very practical ways to solve them without breaking any rules, maintaining valid systems, or disrupting operations.
“The pharmaceutical sector has experienced a phenomenal speed of adoption of automation and Industry 4.0 technologies, moving beyond repetitive tasks and enabling smarter quality systems.”
— Parag Swadia, CEO, Otsuka Pharmaceuticals
Key Digital Adoption Challenges Across Pharma and Biotech
The following table summarizes the most common barriers faced during digital initiatives.
| Challenge | Operational Impact | Digital Solution |
| Legacy IT systems | Data silos | Integration layers |
| Compliance complexity | Slow approvals | AI validation tools |
| Fragmented data | Limited insights | Unified platforms |
| Workforce resistance | Low adoption | Role training |
| Skill gaps | Underused tools | Capability programs |
Outdated infrastructure and disconnected departments create inefficiencies that slow Pharma 4.0 progress. A focused biotech digital strategy addresses these bottlenecks through integration, training, and governance aligned with business outcomes.
| Did you know? |
| Over 70% of pharma companies report that legacy IT systems are the biggest barrier to digital transformation, causing data silos and slowing innovation. |
Compliance Pressure: The Barrier to Innovation
Life sciences companies operate under frameworks like GxP and Part 11, which insist on complete traceability. Each digital tool should support audit trails and validation logs. This ties experimentation with automation and AI initiatives. But now, the requirements embed in workflows from Regulatory AI Compliance tools, which cut manual effort and speed up approvals.
Compliance integrated into technology architecture enables safer digital transformation in life sciences. AI-driven documentation cuts down audit stress and human errors. This gives teams confidence to go ahead with adopting new platforms without fearing regulatory setbacks.
Data Silos and Interoperability Issues
Clinical, quality, and manufacturing data reside in different systems, making real-time viewing by all decision-makers difficult. This results in a fragmented system that slows down collaboration and makes it impossible to take urgent actions across departments. Interconnectedness is key to the success of digital transformation life sciences. API-led integration enables various systems to communicate with each other without necessarily changing the existing platforms, which already have the rules set.
A practical biotech digital strategy focuses on fixing data-sharing issues before adding new tools: when systems share data easily, teams can work better with each other and see what is happening more clearly. This boosts efficiency while keeping everything in line with regulations and causes less disruption to current workflows.
Workforce Resistance and Change Management
The laboratory teams and quality teams rely a lot on the defined documentation and validation processes, making the newly created digital platforms seem complex and risky. The Pharma 4.0 implementation requires organizations to make sure digital tools seamlessly integrate with daily processes and operations, requiring minimal behavioral changes. In this way, user familiarity and trust are created.
Human-centric planning enhances digital transformation in life sciences projects. Role-based training enables employees to embrace digital changes more confidently through good communication and digital champions within the organization. Understanding and recognizing the contributions of digital transformation to their roles encourages employees to embrace these changes more confidently.
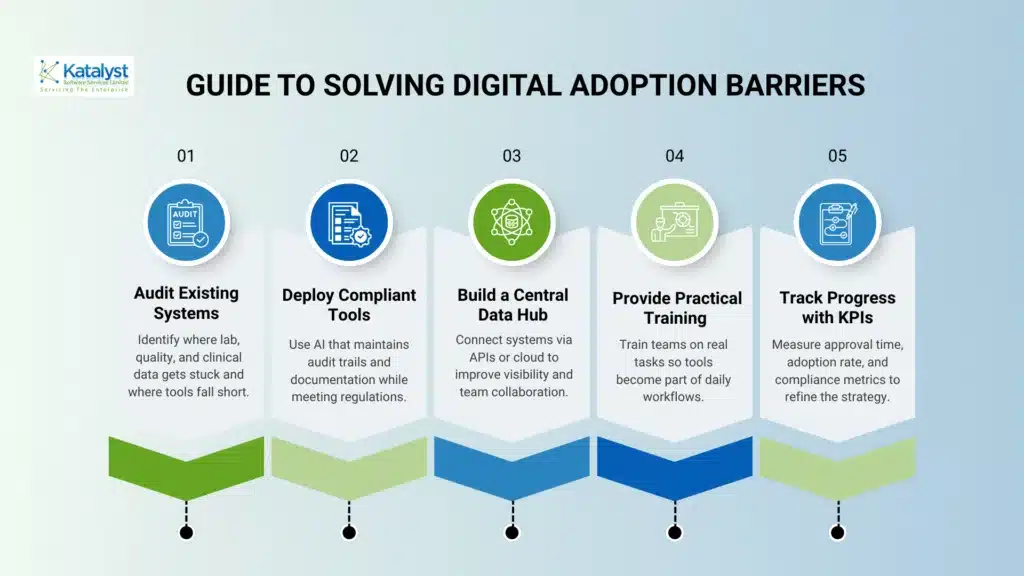
 Step-by-Step Guide to Solving Digital Adoption Barriers
Step-by-Step Guide to Solving Digital Adoption Barriers
A simple plan is needed to makeDigital transformation goals life sciences happen.
Step 1:
Check old systems and connection issues: Look at current lab, quality, and clinical tools to find where data is stuck and where systems fall short. This helps create a clear path for Digital transformation without any complication.
Step 2:
Use AI that meets the rules: Put in AI tools that keep track of audits and documents. This makes sure that new ideas are safe and follow the rules.
Step 3:
Make a central data system: Connect systems with APIs or cloud services to enhance visibility and improve your biotech digital plan across teams.
Step 4:
Offer specific digital training: Train teams with real tasks, so they see how tools help with Digital transformation in everyday work.
Step 5:
Monitor progress with key measures: Track approval times, usage rates, and rule-following numbers to keep improving your biotech digital strategy.
This step-by-step method lowers risks and helps modernize teams steadily.
How AI and Automation Accelerate Pharma 4.0
AI assists in streamlining paperwork. It enhances the efficiency of quality checks. It streamlines batch record checking, problem-solving, and release processes. Companies that employ Pharma 4.0 experience faster releases.
AI additionally helps with Regulatory AI compliance by keeping track of all things in real-time, ensuring that everything could be traced during the course of production. Furthermore, automated checks help reduce the repetition of work so that quality teams can focus more on critical decisions. Firms that employ these technologies report improved confidence in running their operations with compliance to regulations, as well as an improvement in the overall manufacturing process.
| Did you know? |
| Over 72% of pharmaceutical companies are actively exploring machine learning and automation tools to improve quality control and operational efficiency. |
Measurable Outcomes of Successful Digital Adoption
The table below highlights the impact of structured digital programs.
| Outcome | Improvement |
| Approval timelines | Faster processing |
| Operational visibility | Better coordination |
| Documentation effort | Reduced workload |
| Adoption rates | Higher engagement |
| Compliance errors | Lower risk |
These improvements reflect the value of Digital transformation life sciences initiatives supported by a strong biotech digital strategy.
The Future of Digital-First Life Sciences
Life sciences are moving towards connected, data-driven ecosystems where systems, people, and processes align. Early investment in digital transformation in life sciences provides seamless collaboration, traceability, and greater operational efficiency when dealing with increasing data from laboratories and clinical systems. This step-by-step approach delivers digital maturity without disrupting validated workflows or regulatory commitments.
A long-term biotech digital strategy focused on interoperability and scalable architecture prepares firms for evolving regulations and cross-functional collaboration. Digitally mature organizations respond quicker to compliance needs and work easier with their partners and regulators, considering digital adoption as continuous evolution, not a point of upgrade.
Katalyst’s Role in Life Sciences Modernization
Katalyst Technologies assists in the modernization of life sciences organizations through compliance-first approaches that prioritize validated environments. This approach in the digital transformation life sciences industry allows the implementation of the latest technologies with minimal disruptions in the compatibility of the technologies with existing laboratory operations, clinical environments, as well as quality systems.
Their knowledge equally enhances the strength of Regulatory AI Compliance by infusing the features of traceability, documentation, and audits within digital projects. This synergy between governance and innovation is aimed at accelerating digital maturity in the regulated sphere.
Turning Digital Challenges into Compliant Growth
Digital adoption challenges faced by life sciences organizations can be addressed through structured planning, interoperability, and compliance-conscious technologies. Organizations embracing Digital transformation life sciences through a governance-conscious approach can benefit from improved approvals, efficiency, and data integrity.
Schedule a demo with Katalyst Technologies as it enables your path to modernization with AI-driven and compliant solutions that cater specifically to the needs of the life sciences industry.
Frequently Asked Questions
Your most common questions, answered with precision and insight